2-Butanone
- CAS No.
- 78-93-3
- Chemical Name:
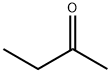
- 2-Butanone
- Synonyms
- MEK;METHYL ETHYL KETONE;BUTANONE;Butan-2-one;ETHYL METHYL KETONE;2-Butanone,99%;2-Butanon;METHYL ETHYL KETONE (MEK)(BUTANONE);Butanon;MEK = 2-BUTANONE
- CBNumber:
- CB4854386
- Molecular Formula:
- C4H8O
- Molecular Weight:
- 72.11
- MOL File:
- 78-93-3.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/3/14 15:18:26
| Melting point | -87 °C (lit.) |
|---|---|
| Boiling point | 80 °C (lit.) |
| Density | 0.805 g/mL at 25 °C (lit.) |
| vapor density | 2.49 (vs air) |
| vapor pressure | 71 mm Hg ( 20 °C) |
| refractive index |
n |
| FEMA | 2170 | 2-BUTANONE |
| Flash point | 26 °F |
| storage temp. | 2-8°C |
| solubility | Miscible with acetone, ethanol, benzene, ether (U.S. EPA, 1985), and many other solvents, particularly ketones and aldehydes |
| pka | 14.7 (quoted, Riddick et al., 1986) |
| form | Solution |
| color | Colorless |
| Relative polarity | 0.327 |
| Odor | Sweet/sharp odor detectable at 2 to 85 ppm (mean = 16 ppm) |
| PH | pH(1+4, 25℃):6.0~7.0 |
| explosive limit | 1.8-11.5%(V) |
| Odor Threshold | 0.44ppm |
| Odor Type | ethereal |
| Water Solubility | 290 g/L (20 ºC) |
| Merck | 14,6072 |
| JECFA Number | 278 |
| BRN | 741880 |
| Henry's Law Constant | 23.0 at 50.00 °C, 34.1 at 60.00 °C, 50.6 at 70.00 °C, 70.4 at 80.00 °C (headspace-GC, Hovorka et al., 2002) |
| Exposure limits | TLV-TWA, PEL 590 mg/m3 (200 ppm) (ACGIH, OSHA); STEL 885 mg/m3 (300 ppm) (ACGIH); IDLH 3000 ppm (NIOSH). |
| Dielectric constant | 18.5(20℃) |
| Stability | Stable. Highly flammable. Incompatible with oxidizing agents, bases, strong reducing agents. Protect from moisture. |
| LogP | 0.3 at 40℃ |
| CAS DataBase Reference | 78-93-3(CAS DataBase Reference) |
| NIST Chemistry Reference | 2-Butanone(78-93-3) |
| EPA Substance Registry System | Methyl ethyl ketone (78-93-3) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS02,GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H225-H319-H336 | |||||||||
| Precautionary statements | P210-P305+P351+P338 | |||||||||
| Hazard Codes | F,Xi,T | |||||||||
| Risk Statements | 11-36-66-67-39/23/24/25-23/24/25 | |||||||||
| Safety Statements | 9-16-45-36/37 | |||||||||
| OEB | A | |||||||||
| OEL | TWA: 200 ppm (590 mg/m3), STEL: 300 ppm (885 mg/m3) | |||||||||
| RIDADR | UN 1193 3/PG 2 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | EL6475000 | |||||||||
| Autoignition Temperature | 516 °C | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 3 | |||||||||
| PackingGroup | II | |||||||||
| HS Code | 29141200 | |||||||||
| Toxicity | LD50 orally in rats: 6.86 ml/kg (Smyth) | |||||||||
| IDLA | 3,000 ppm | |||||||||
| NFPA 704 |
|
2-Butanone price More Price(5)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| ottokemi | E 1619 | Ethyl methyl ketone, for HPLC 99.7% | 78-93-3 | 500mL | ₹891 | 2022-05-26 | Buy |
| ottokemi | E 1617 | Ethyl methyl ketone , GR 99%+ | 78-93-3 | 500mL | ₹891 | 2022-05-26 | Buy |
| ottokemi | E 1617 | Ethyl methyl ketone , GR 99%+ | 78-93-3 | 5lt | ₹6903 | 2022-05-26 | Buy |
| ottokemi | E 1615 | Ethyl methyl ketone 99% | 78-93-3 | 500mL | ₹792 | 2022-05-26 | Buy |
| ottokemi | E 1615 | Ethyl methyl ketone 99% | 78-93-3 | 5lt | ₹6201 | 2022-05-26 | Buy |
2-Butanone Chemical Properties,Uses,Production
Description
2-Butanone is a stable, highly flammable chemical. It is incompatible with oxidising agents, bases, and strong reducing agents. It is a colourless liquid with a sharp, sweet odour. 2-Butanone is produced in large quantities. It is used as a solvent and nearly half of its use is in paints and other coatings because it will quickly evaporate into the air and it dissolves many substances. It is also used in glues and as a cleaning agent.
Chemical Properties
Methyl ethyl ketone (MEK) is a colorless liquid with an odor that has been described as moderately sharp, fragrant, peppermint, or acetone like. It soluble in water up to 28% by weight and is miscible with many other organic solvents. The lower explosive limit is 1.4% and the upper explosive limit is 11.4%. Methyl ethyl ketone may be incompatible with strong oxidizers, amines, ammonia, inorganic acids, caustics, isocyanates, and pyridines. When used industrially, methyl ethyl ketone must be handled with caution, as it is a Class lB flammable liquid NIOSH (2010).
Physical properties
2-Butanone is a clear, colorless, volatile, very flammable liquid with a sweet, mint or acetone-like odor. Odor threshold concentration is 10.0 ppmv (Leonardos et al., 1969). Experimentally determined detection and recognition odor threshold concentrations were 5.8 mg/m3 (2.0 ppmv) and 16 mg/m3 (5.4 ppmv), respectively (Hellman and Small, 1974). It is incompatible with oxidizing agents, bases, and strong reducing agents. It is a colorless liquid with a sharp, sweet odor. 2-Butanone is produced in large quantities. It is used as a solvent and nearly half of its use is in paints and other coatings because it quickly evaporates into the air and it dissolves many substances. It is also used in glues and as a cleaning agent.
Occurrence
Reported found as an impurity among products from the dry distillation of wood and in the oil (extracted with ether) of black tea; it is also present in coffee, cheese, bread, some citrus oils and some other natural products (grape, raspberry).
Uses
2-Butanone is an eye irritant that has been used as a water soluble photoinitiator for the photopolymerization of methacrylic acid (MAA). As solvent; in the surface coating industry; manufacture of smokeless powder; colorless synthetic resins.
Production Methods
Methyl ethyl ketone is commercially manufactured from nbutene in a metal-catalyzed hydrogenation reaction that proceeds through the intermediate formation of 2-butanol . A second method of synthesis involves the liquid- phase oxidation of n-butane with the formation of acetic acid as a coproduct.
Preparation
By catalytic dehydrogenation of secondary butyl alcohol; by dehydration of butane-2,3-diol by refluxing with 25% aqueous H2SO4. Industrially, it is also prepared by controlled oxidation of butane, by dry distillation of calcium acetate and calcium propionate, or by refluxing methyl acetoacetate and diluted H2SO4.
Definition
ChEBI: A dialkyl ketone that is a four-carbon ketone carrying a single keto- group at position C-2.
General Description
Methyl ethyl ketone (MEK) is a colourless liquid with a sweet and sharp odour. It is soluble in alcohol, ether, acetone benzene, and water. It is a solvent often found in mixtures with acetone, ethyl acetate, n-hexane, toluene, or alcohols. It has applications in the surface coating industry and in the de-waxing of lubricating oils. MEK is used in the manufacture of colourless synthetic resins, artificial leather, rubbers, lacquers, varnishes, and glues.
Air & Water Reactions
Highly flammable. Slightly soluble in water.
Hazard
Flammable, dangerous fire risk. Toxic by ingestion.
Health Hazard
Occupational workers are exposed to 2-butanone by breathing contaminated air in workplaces associated with the production or use of paints, glues, coatings, or cleaning agents. Prolonged exposures to 2-butanone cause symptoms of poisoning such as cough, dizziness, drowsiness, headache, nausea, vomiting, dermatitis, irritation of the nose, throat, skin, and eyes and at very high levels cause drooping eyelids, uncoordinated muscle movements, loss of consciousness, and birth defects. Chronic inhalation studies in animals have reported slight neurological, liver, kidney, and respiratory effects. However, information on the chronic (long-term) effects of 2-butanone (methyl ethyl ketone) in humans is limited.
Fire Hazard
Flammable/combustible material. May be ignited by heat, sparks or flames. Vapors may form explosive mixtures with air. Vapors may travel to source of ignition and flash back. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapor explosion hazard indoors, outdoors or in sewers. Runoff to sewer may create fire or explosion hazard. Containers may explode when heated. Many liquids are lighter than water.
Flammability and Explosibility
Methyl ethyl ketone is extremely flammable (NFPA rating = 3), and its vapor can travel a considerable distance to an ignition source and "flash back." MEK vapor forms explosive mixtures with air at concentrations of 1.9 to 11% (by volume). Carbon dioxide or dry chemical extinguishers should be used for MEK fires.
Chemical Reactivity
Reactivity with Water No reaction; Reactivity with Common Materials: No reaction; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Not pertinent; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent.
Potential Exposure
MEK is used as a solvent in nitrocellulose coating and vinyl film manufacture; in smokeless powder manufacture; in cements and adhesives and in the dewaxing of lubricating oils. It is also an intermediate in drug manufacture
Carcinogenicity
Although MEK has not been specifically examined in a rodent 2-year bioassay, there is little to suggest that the material is carcinogenic. When used as a delivery vehicle in a dermal carcinogenicity bioassay for organic sulfur compounds, Horton et al. found that the application of benzyl disulfide or phenylbenzylthiophene in a 25–29% solution of MEK in dodecylbenzene together failed to increase the incidence of benign skin papillomas in male C3H/HeJmice. The mice used in the experiments were treated twice a week for 52 weeks with the MEKcontaining test solution.
storage
2-Butanone should be protected from moisture.
Shipping
UN1193 Methyl ethyl ketone or Ethyl methyl ketone, Hazard Class: 3; Labels: 3-Flammable liquid.
Incompatibilities
May form explosive mixture with air. Violent reaction with strong oxidizers, amines, ammonia, inorganic acids; caustics, isocyanates, pyridines. Incompatible with potassium tert-butoxide, 2-propanol, chlorosulfonic acid; oleum. Attacks some plastics. Ketones are incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides, nitrated amines, azo, diazo, azido compounds, carbamates, organic cyanates
Waste Disposal
Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform to EPA regulations governing storage, transportation, treatment, and waste disposal. Incineration
Precautions
2-Butanone vapor and air mixtures are explosive. It reacts violently with strong oxidants and inorganic acids causing fi re and explosion hazard.
2-Butanone Preparation Products And Raw materials
Raw materials
1of3
chevron_rightPreparation Products
1of8
chevron_right78-93-3(2-Butanone)Related Search:
1of4
chevron_right




