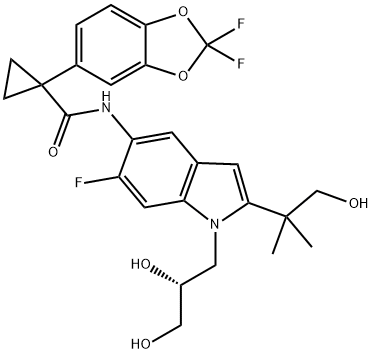
Tezacaftor
|
- ₹0
- Product name: Tezacaftor
- CAS: 1152311-62-0
- MF: C26H27F3N2O6
- MW: 520.5
- EINECS:
- MDL Number:MFCD23106064
- Synonyms:VX-661;VX-661/VX661;1-(2,2-Difluoro-1,3-benzodioxol-5-yl)-N-[1-[(2R)-2,3-dihydroxypropyl]-6-fluoro-2-(2-hydroxy-1,1-dimethylethyl)-1H-indol-5-yl]-cyclopropanecarboxamide;Cyclopropanecarboxamide, 1-(2,2-difluoro-1,3-benzodioxol-5-yl)-N-[1-[(2R)-2,3-dihydroxypropyl]-6-fluoro-2-(2-hydroxy-1,1-dimethylethyl)-1H-indol-5-yl]-;1-(2,2-Difluoro-1,3-benzodioxol-5-yl)-N-[1-[(2R)-2,3-dihydroxypropyl]-6-fluoro-2-(2-hydroxy-1,1-dimethylethyl)-1H-indol-5-yl]-cyclopropanecarboxamide VX661;(R)-1-(2,2-Difluorobenzo[d][1,3]dioxol-5-yl)-N-(1-(2,3-dihydroxypropyl)-6-fluoro-2-(1-hydroxy-;CS-956;VX661/Tezacaftor
| Manufacturer | Product number | Product description | Packaging | Price | Updated | Buy |
|---|
Properties
Boiling point :610.8±55.0 °C(Predicted)
Density :1.49±0.1 g/cm3(Predicted)
storage temp. :Store at -20°C , stored under nitrogen
solubility :≥21.8 mg/mL in DMSO; insoluble in EtOH; ≥24.3 mg/mL in H2O
form :solid
pka :13.99±0.20(Predicted)
color :White to yellow
InChIKey :MJUVRTYWUMPBTR-MRXNPFEDSA-N
SMILES :C1(C2=CC=C3OC(F)(F)OC3=C2)(C(NC2C(F)=CC3=C(C=2)C=C(C(C)(C)CO)N3C[C@@H](O)CO)=O)CC1
Density :1.49±0.1 g/cm3(Predicted)
storage temp. :Store at -20°C , stored under nitrogen
solubility :≥21.8 mg/mL in DMSO; insoluble in EtOH; ≥24.3 mg/mL in H2O
form :solid
pka :13.99±0.20(Predicted)
color :White to yellow
InChIKey :MJUVRTYWUMPBTR-MRXNPFEDSA-N
SMILES :C1(C2=CC=C3OC(F)(F)OC3=C2)(C(NC2C(F)=CC3=C(C=2)C=C(C(C)(C)CO)N3C[C@@H](O)CO)=O)CC1
Safety Information
| Symbol(GHS): |

|
|||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Signal word: | Warning | |||||||||||||||||||||||||||||||||||
| Hazard statements: |
|
|||||||||||||||||||||||||||||||||||
| Precautionary statements: |
|
Description
Tezacaftor (VX-661) is an oral medication for cystic fibrosis (CF) developed by Vertex Pharmaceuticals. In a combination therapy formulation called Symdeko (tezacaftor plus ivacaftor), it is approved in the U.S. and Canada for CF patients, ages 12 and older, who have two copies of the F508del mutation in the CFTR gene and one minimal function mutation. Symdeko is approved and marketed in the EU as Symkevi.Tezacaftor is not approved as a stand-alone treatment, but as part of a combination therapy.More related product prices
Ataluren (PTC124) CFTRinh-172 Lumacaftor (VX-809) VX-770 ABT 737 918505-84-7 VX 702 896720-20-0 VX-222 VX-745 Dibenzothiophene Thianaphthene BENZO[A]PYRENE (S)-1-((S)-2-(4-amino-3-chlorobenzamido)-3,3-dimethylbutanoyl)-N-((2R,3S)-2-ethoxy-5-oxotetrahydrofuran-3-yl)pyrrolidine-2-carboxamideRelated product price
- Ataluren (PTC124)
₹18830 - CFTRinh-172
₹16390-66974.28 - VX-770
₹22390






