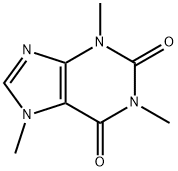
Caffeine
- CAS No.
- 58-08-2
- Chemical Name:
- Caffeine
- Synonyms
- CAFFEIN;Caffine;COFFEIN;1,3,7-Trimethyl-3,7-dihydro-1H-purine-2,6-dione;COFFEINE;1,3,7-triMethyl-1H-purine-2,6(3H,7H)-dione;1H-Purine-2,6-dione, 3,7-dihydro-1,3,7-trimethyl-;KFY;caffeina;Natural Caffeine
- CBNumber:
- CB0202769
- Molecular Formula:
- C8H10N4O2
- Molecular Weight:
- 194.19
- MOL File:
- 58-08-2.mol
- Modify Date:
- 2023/6/30 15:45:59
| Melting point | 234-236.5 °C(lit.) |
|---|---|
| Boiling point | 178°C |
| Density | 1.23 |
| refractive index | 1.6590 (estimate) |
| FEMA | 2224 | CAFFEINE |
| Flash point | 178°C |
| storage temp. | 2-8°C |
| solubility | Sparingly soluble in water, freely soluble in boiling water, slightly soluble in ethanol (96 per cent). It dissolves in concentrated solutions of alkali benzoates or salicylates. |
| form | Crystals or Crystalline Powder |
| pka | pKa 0.6 (Uncertain) |
| color | Silky white or white |
| PH | pH (10g/l, 25℃) : 5.5~6.5 |
| Odor | at 100.00 %. odorless |
| Odor Type | odorless |
| Water Solubility | 20 g/L (20 ºC) |
| Sublimation | 178 ºC |
| Merck | 14,1636 |
| BRN | 17705 |
| Stability | Stable. Incompatible with strong acids, strong bases, strong oxidizing agents, iodine, silver salts, tannins. Weakly light sensitive in solution. |
| LogP | -0.07 |
| CAS DataBase Reference | 58-08-2(CAS DataBase Reference) |
| IARC | 3 (Vol. 51) 1991 |
| NIST Chemistry Reference | Caffeine(58-08-2) |
| EPA Substance Registry System | Caffeine (58-08-2) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H302 | |||||||||
| Precautionary statements | P301+P312+P330 | |||||||||
| Hazard Codes | Xn,T,F | |||||||||
| Risk Statements | 22-25-39/23/24/25-23/24/25-11 | |||||||||
| Safety Statements | 16-36/37-45-7 | |||||||||
| RIDADR | UN 1544 6.1/PG 3 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | EV6475000 | |||||||||
| F | 10 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 6.1 | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 29393000 | |||||||||
| Toxicity | LD50 orally in mice, hamsters, rats, rabbits (mg/kg): 127, 230, 355, 246 (males); 137, 249, 247, 224 (females) (Palm) | |||||||||
| NFPA 704 |
|
Caffeine price More Price(35)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | W222402 | Caffeine anhydrous, 99%, FCC, FG | 58-08-2 | 1SAMPLE-K | ₹5141.88 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W222402 | Caffeine anhydrous, 99%, FCC, FG | 58-08-2 | 1KG | ₹8411.03 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | PHR1009 | Caffeine Pharmaceutical Secondary Standard; Certified Reference Material | 58-08-2 | 1G | ₹8530.1 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | C8960 | Caffeine BioXtra | 58-08-2 | 250G | ₹17244.23 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | C8960 | Caffeine BioXtra | 58-08-2 | 1KG | ₹31554.88 | 2022-06-14 | Buy |
Caffeine Chemical Properties,Uses,Production
Description
Caffeine is an alkaloid purine belonging to the group of organic compounds called methylxanthines. Pure caffeine is a white, crystalline, bitter-tasting compound. Caffeine is found in a number of plants, principally coffee and tea plants, as well as cola and cacao nuts. In plants, caffeine functions as a natural pesticide to deter insects.
Chemical Properties
Caffeine is the alkaloid 1,3,7-trimethylxanthine. It is one of the xanthine derivatives present up to 1.5% in seeds of coffee (Coffea arabica L.) and up to 5% in the leaves of tea (Camelia sinensis). It is a component of the beverages made from these plants. Caffeine is also a component of chocolate (Theobroma cacao) and the cola nut (Cola acuminata Schott and Endel. and related species), the extract of which is used in cola drinks. It is virtually odorless. Caffeine is added to cola-type beverages for its enhancement of flavor. Subtle and subliminal flavors are widely appreciated by consumers and caffeine has a modifying effect on other components of the beverage. The threshold for detecting the presence of caffeine in liquid foods varies depending on the nature of other substances present, but lies close to the level characteristic of currently produced cola-type beverages. The threshold for detection of caffeine in water has been shown to be 0.0095%; in liquid foods, 0.0184%. In one study, panelists could distinguish a solution containing 0.0058% caffeine from the control. The threshold for detecting taste difference between an aqueous solution of caffeine and a water control was also shown to be 0.005% caffeine and to distinguish bitterness, 0.011% caffeine. In aqueous solutions containing threshold and subthreshold concentrations of caffeine, sucrose, citric acid and salt, all compounds depressed the taste intensity of each other.*
Physical properties
Appearance: odorless silky needle-like crystal or crystal powder with the color of white or a little yellowish green. Solubility: weathering, easily dissolved in water or chloroform and slightly soluble in water, ethanol, or acetone, very slightly dissolved in ether. Melting point: 235–238?°C.
Occurrence
Reported found in coffee and guarana.
History
Runge isolated caff eine from coffee in 1819. Caffeine derives its name from the Kaffa region of Ethiopia. Caffeine comes from the German kaffeine, which in turn is derived from the German word for coffee, kaffee. In 1827, a compound isolated from tea was named theine, but this was eventually shown to be caffeine.
Uses
Caffeine is a bitter, white crystalline xanthine alkaloid that acts as a stimulant drug and a reversible acetylcholinesterase inhibitor. Caffeine is found in varying quantities in the seeds, leaves, and fruit of some plants, where it acts as a natural pesticide that paralyzes and kills certain insects feeding on the plants. In humans, caffeine acts as a central nervous system stimulant, temporarily warding off drowsiness and restoring alertness. Caffeine is a cardiac and respiratory stimulant; diuretic. Caffeine is toxic at sufficiently high doses.
Indications
This product is included in the Pharmacopoeia of the People’s Republic of China (2015), the British Pharmacopoeia (2017), the United States Pharmacopeia (40), the Japanese Pharmacopoeia (17th ed.), the European Pharmacopoeia (9.0th ed.), the Indian Pharmacopoeia (2010), and the International Pharmacopoeia (5th ed.). Commonly used dosage forms of caffeine include tablet, powder, and injection. Mainly used dosage forms in the market include caffeine citrate tablets, amidopyrine caffeine tablets, amidopyrine caffeine, children acetaminophen aspirin caffeine tablets, ergotamine caffeine tablets, caffeine sodium benzoate injection, cafe bromine agent, etc.
Definition
ChEBI: A trimethylxanthine in which the three methyl groups are located at positions 1, 3, and 7. A purine alkaloid that occurs naturally in tea and coffee.
General Description
Odorless white powder or white glistening needles, usually melted together. Bitter taste. Solutions in water are neutral to litmus. Odorless.
Air & Water Reactions
Efflorescent in air. Water soluble.
Reactivity Profile
Caffeine may be hygroscopic. Aqueous solutions (1.12 mg/mL) are stable for three weeks at 41° F if protected from light. In normal room lighting and at room temperature, solutions are stable for 3 days. Solutions of Caffeine in water, DMSO, 95% ethanol or acetone should be stable for 24 hours under normal lab conditions. REACTIVITY: Caffeine may react with strong oxidizing agents. Caffeine is also incompatible with iodine, silver salts and tannins. Caffeine is a very weak base. Caffeine is decomposed by strong solutions of caustic alkalis.
Hazard
One grain or more is toxic, 200 μg/m L has been found to inhibit activity of the enzyme DNA polymerase. Use in soft drinks not to exceed 0.02%. Questionable carcinogen.
Health Hazard
Caffeine is a stimulant of the central nervoussystem. It eliminates fatigue and drowsiness. However, high doses cause gastrointestinal motility, restlessness, sleeplessness,nervousness, and tremor. Acute poisoningeffects include nausea, vomiting, headache,excitability, tremor, and sometimes, convulsive coma. Other symptoms may be respiratory depression, muscle contraction, distortedperception, and hallucination. Ingestion of15–20 g may be fatal to humans.
LD50 value, oral (mice): 127 mg/kg
LD50 value, oral (rabbits): 224 mg/kg
Animal studies indicate that caffeine athigh doses produces adverse reproductiveeffects, causing developmental abnormalities. It tested negative in the histidine reversion–Ames and TRP reversion tests.
Fire Hazard
Flash point data for Caffeine are not available; however, Caffeine is probably combustible.
Biological Activity
Central nervous system stimulant. Antagonist at A 1 and A 2A adenosine receptors and inhibitor of cyclic nucleotide phosphodiesterases. Mobilises calcium from intracellular stores and inhibits benzodiazepine binding to GABA receptors.
Clinical Use
The commonly used clinical preparations include caffeine sodium benzoate and ergotamine caffeine. The preparation of caffeine sodium benzoate (injection) is constituted of 0.12?g/ml of caffeine, 0.13?g/ml of sodium benzoate, and cafe bromine mixture (oral liquid). Clinically, it can be used for migraine headaches, cerebral artery dilated headache, or headache caused by histamine. However, it is invalid in the prevention of headaches. The adverse reactions include nausea, vomiting, abdominal pain, and fatigue. Other common symptoms include numbness and tingling of the hands, toes, and face and swelling of the foot and lower limb. Overdose causes severe poisoning, mental disorder, ataxia, convulsions, gray chills of the hand and foot, sensory disturbance, and even death due to coma and respiratory paralysis. Caffeine citrate preparation, including injection and oral solution, is the only internationally approved drug for the treatment of premature infant apnea.
Safety Profile
A human poison by ingestion. An experimental poison by ingestion, subcutaneous, intraperitoneal, intramuscular, rectal, and intravenous routes. Human systemic effects: ataxia, blood pressure elevation, change in heart rate, changes in tubules, convulsions or effect on seizure threshold, dtarrhea, distorted perceptions, hallucinations, hypermotility, muscle contraction, musculoskeletal tumors, nausea or vomiting, toxic psychosis, tremors. A human teratogen causing developmental abnormalities of the craniofacial and musculoskeletal systems, pregnancy termination (abortion), and stillbirth. Human maternal effects include an unspecified effect on labor or chddbirth. Human mutation data reported. An experimental teratogen. Other experimental reproductive effects. Questionable carcinogen with experimental carcinogenic data. Large doses (above 1.0 g> cause palpitation, excitement, insomnia, dtzziness, headache, and vomiting. Continued excessive use of caffeine in tea or coffee may lead to digestive disturbances, constipation, palpitations, shortness of breath, and depressed mental states. It is also implicated in cardiac disorders under those condttions. When heated to decomposition it emits toxic fumes of NOx
Environmental Fate
Caffeine can have profound effects on the cardiovascular
system. At least four mechanisms have been proposed for the
pro-arrhythmic potential of caffeine in overdose. First,
caffeine increases circulating catecholamines. Second, caffeine
inhibits phosphodiesterase. Increased circulating catecholamines
after caffeine overdose increase b1-receptor stimulation.
Stimulation of b1-receptors increases intracellular cAMP
by G protein stimulation of adenylate cyclase. The activity of
cAMP is prolonged due to its decreased metabolism as
phosphodiesterase is inhibited by caffeine. Subsequently,
b1-receptor effects are exaggerated and tachydysrhythmias are
induced. Third, caffeine increases myocardial intracellular calcium. Caffeine both induces release of calcium from the
sarcoplasmic reticulumand blocks calcium’s reuptake into the
sarcoplasmic reticulum. This resulting increase in cytosolic
calcium may provoke dysrhythmias. Fourth, caffeine blocks
cardiac adenosine receptors, which have been shown to be
antiarrhythmic.
The hypotension that has been noted with overdoses of
caffeine is due primarily to two mechanisms. First, caffeineinduced
tachydysrhythmias lead to inadequate filling of the
heart and subsequent decrease in cardiac output. Second,
caffeine augments β2-effects and causes subsequent vasodilation
with resulting hypotension.
Purification Methods
Caffeine crystallises from water or absolute EtOH. [Beilstein 26 III/IV 2338.]
Caffeine Preparation Products And Raw materials
Raw materials
1of4
chevron_rightPreparation Products
Related articles
- General Introduction of Caffeine
- Caffeine has recently become popular as a component of topical cosmeceuticals due to its well-known biological activity. It ha....
- Mar 3,2022
58-08-2(Caffeine)Related Search:
1of4
chevron_right




