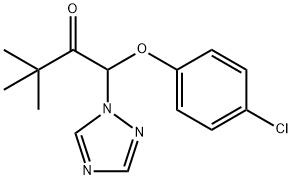
Triadimefon
- CAS No.
- 43121-43-3
- Chemical Name:
- Triadimefon
- Synonyms
- Tridimefon;Triadimefone;TRIADIMEPHON;TriadiMefon Standard;ROFON;NUREX;OTRIA;DAYTON;ACIZOL;Amiral
- CBNumber:
- CB7426290
- Molecular Formula:
- C14H16ClN3O2
- Molecular Weight:
- 293.75
- MOL File:
- 43121-43-3.mol
- Modify Date:
- 2024/8/2 16:47:38
| Melting point | 82°C |
|---|---|
| Boiling point | 441.9±55.0 °C(Predicted) |
| Density | 1.2200 |
| vapor pressure | 2 x l0-5 Pa (20 °C) |
| refractive index | 1.5388 (estimate) |
| Flash point | 11 °C |
| storage temp. | APPROX 4°C |
| solubility | DMF: 30 mg/ml; DMSO: 30 mg/ml; DMSO:PBS(pH7.2) (1:1): 0.5 mg/ml; Ethanol: 10 mg/ml |
| form | Solid |
| pka | 1.41±0.11(Predicted) |
| color | White to off-white |
| Water Solubility | 0.026 g/100 mL |
| Merck | 13,9666 |
| BRN | 619231 |
| NIST Chemistry Reference | 2-Butanone, 1-(4-chlorophenoxy)-3,3-dimethyl-1-(1h-1,2,4-triazol-1-yl)-(43121-43-3) |
| EPA Substance Registry System | Triadimefon (43121-43-3) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS06,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H302-H317-H330-H410 | |||||||||
| Precautionary statements | P260-P273-P280-P301+P312-P302+P352-P304+P340+P310 | |||||||||
| Hazard Codes | Xn,N,T,F | |||||||||
| Risk Statements | 22-43-51/53-39/23/24/25-23/24/25-11 | |||||||||
| Safety Statements | 24-37-61-45-36/37 | |||||||||
| RIDADR | 2588 | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | EL7100000 | |||||||||
| HazardClass | 6.1(b) | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 29339900 | |||||||||
| Toxicity | LD50 in male, female rats (mg/kg): 568, 363 orally (Michel, Pourcharesse) | |||||||||
| NFPA 704 |
|
Triadimefon Chemical Properties,Uses,Production
Description
Triadimefon has been a widely used fungicide on crops and nonfood products since the early 1970s. The metabolite triadimenol is also active and is registered separately for use as seed treatment. Triadimenol has a broad regulatory toxicology database, but its toxicity is considered to be encompassed in that of triadimefon and therefore the same study was used by the United States Environmental Protection Agency (US EPA) in establishing regulatory levels for both pesticides. In nontarget species, dopaminergic neurotoxicity is the primary effect, but with chronic exposures its toxicities include hepatic, carcinogenic, developmental, and reproductive effects.
Uses
Triadimefon is an triazole fungicide is used for the management of mango powdery mildew in South Gujarat.
General Description
Colorless to pale yellow crystalline solid with a slight odor.
Air & Water Reactions
Insoluble in water.
Reactivity Profile
Triadimefon is incompatible with strong oxidizing agents and acids. Reacts with acid halides and anhydrides. Also reacts with most active hydrogen compounds .
Fire Hazard
Flash point data for Triadimefon are not available; however, Triadimefon is probably combustible.
Agricultural Uses
Fungicide: Triadimefon is a systemic fungicide that is used to control powdery mildews, rusts, and other fungi on coffee, seed grasses, cereals, fruits, grapes, vegetables, vines, pineapple, sugar cane, sugar beets, turf, shrubs, and trees. Not approved for use in EU countries. Registered for use in the U.S. U.S. Maximum Allowable Residue Levels for Triadimefon
Trade name
ACCOST®; ACIZOL®; AMIRAL®; BAY® 6681-F; BAYLETON®; BAY®-MEB-6447; BAYER® 6681-F; BAYER® MEB-6447; MEB 6447®; PRO-TEK®; ROFON®
Pharmacology
Triadimefon (36) and its alcohol analog triadimenol (37) have been intensively investigated to determine the influence of their enantiomeric difference on fungicidal activity. Between stereoisomeric triadimefon, no significant difference is observed in their fungicidal activity. However, triadimenol, which shows a much higher fungicidal activity than triadimefon, exhibits a clear stereochemistry-dependent activity difference. Greater fungicidal activity is possessed by the (1S, 2R)-isomer (28).
Safety Profile
Poison by ingestion. Mutation data reported. When heated to decomposition it emits very toxic fumes of Cland NOx. See also KETONES.
Environmental Fate
Soil. In a culture study, the microorganism Aspergillus niger degraded 32% of tri-
adimefon to triadimenol after 5 days (Clark et al., 1978).
Plant. In soils and plants, triadimefon degrades to triadimenol (Clark et al., 1978;
Rouchaud et al., 1981). In barley plants, triadimefon was metabolized to triadimenol and
p-chlorophenol (Rouchaud et al., 1981; Rouchaud, 1982). In the grains an
Photolytic. When triadimefon was subjected to UV light for one week, p-chlorophenol,
4-chlorophenyl methyl carbamate and a 1,2,4-triazole formed as products (Clark et al.,
1978).
Metabolic pathway
Enzymic reduction of triadimefon is an important pathway in plants, soils and fungi and may be regarded as an activation process, which produces fungicidally active triadimenol. Two diastereoisomers of triadimenol, A and B [( 1RS,2SR)-l-(4-chlorophenoxy)-3,3-dimethyl-1-(1H-1,2,4-triazol-1-yl) butan-2-ol is referred to as diastereoisomer A; 1RS,2RS- is referred to as diastereoisomer β], are produced in different amounts by plants and fungi and the proportions may differ within the plant. Similar metabolic pathways are followed in mammals where reduction of the keto group yields triadimenol as the principal metabolite and oxidation of the butyl group gives alcohol and carboxylic acid derivatives.
Triadimefon Preparation Products And Raw materials
Raw materials
1of6
chevron_rightPreparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| Parijat Industries India Private Limited | 08048372460Ext 265 | Delhi, India | 19 | 58 | Inquiry |
| Sonachi Industries Ltd | 09870760066 | Mumbai, India | 2 | 58 | Inquiry |
| Hebei Guanlang Biotechnology Co,.LTD | +86-19930503253; +8619930503252 | China | 5835 | 58 | Inquiry |
| Hebei Jingbo New Material Technology Co., Ltd | +8619931165850 | China | 1000 | 58 | Inquiry |
| Hebei Saisier Technology Co., LTD | +86-18400010335 +86-18034520335 | China | 989 | 58 | Inquiry |
| Hebei Longbang Technology Co., LTD | +86-18032476855 +86-18032476855 | China | 948 | 58 | Inquiry |
| HebeiShuoshengImportandExportco.,Ltd | +undefined18732180219 | China | 207 | 58 | Inquiry |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | China | 21669 | 55 | Inquiry |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 | China | 2989 | 55 | Inquiry |
Related articles
- Triadimefon: Balancing Agricultural Benefits with Health Hazards in Plant Growth Regulation
- Triadimefon controls plant diseases and promotes growth, but presents health hazards like skin irritation. Safety measures are....
- Feb 27,2024
- What is triadimefon used for?
- Triadimefon is a systemic fungicide in the triazole family of chemicals, which are also called triazole or imidazole-containin....
- May 22,2023
43121-43-3(Triadimefon)Related Search:
1of4
chevron_right




