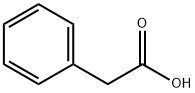
Phenylacetic acid
- CAS No.
- 103-82-2
- Chemical Name:
- Phenylacetic acid
- Synonyms
- 2-phenylacetic acid;BENZENEACETIC ACID;phenylacetic;FEMA 2878;PhenylaceticAcidC8H8O2;BENZYLFORMIC ACID;Phenylacetic acid, 98.50%;Toluylicacid;L-PHENYLALAIN;α-Toluic acid
- CBNumber:
- CB9699850
- Molecular Formula:
- C8H8O2
- Molecular Weight:
- 136.15
- MOL File:
- 103-82-2.mol
- MSDS File:
- SDS
- Modify Date:
- 2023/11/28 16:31:44
| Melting point | 76-78 °C(lit.) |
|---|---|
| Boiling point | 265 °C(lit.) |
| Density | 1.081 g/mL at 25 °C(lit.) |
| vapor density | ~4 (vs air) |
| vapor pressure | 1 mm Hg ( 97 °C) |
| FEMA | 2878 | PHENYLACETIC ACID |
| refractive index | 1.5120 (estimate) |
| Flash point | 132°C |
| storage temp. | Store at RT. |
| solubility | DMF: 1 mg/ml; PBS (pH 7.2): 10 mg/ml |
| pka | 4.28(at 18℃) |
| Specific Gravity | 1.081 |
| color | Leaflets on distillation in vac; plates, tablets from pet ether |
| Odor | disagreeable odor of geranium |
| PH | 3.7(1 mM solution);3.17(10 mM solution);2.66(100 mM solution) |
| Odor Type | honey |
| Water Solubility | 15 g/L (20 ºC) |
| Merck | 14,7268 |
| JECFA Number | 1007 |
| BRN | 1099647 |
| Dielectric constant | 3.0(20℃) |
| Stability | Stable. Incompatible with strong oxidizing agents. |
| LogP | 0.811 at 25℃ |
| CAS DataBase Reference | 103-82-2(CAS DataBase Reference) |
| NIST Chemistry Reference | Benzeneacetic acid(103-82-2) |
| EPA Substance Registry System | Phenylacetic acid (103-82-2) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H319 | |||||||||
| Precautionary statements | P305+P351+P338 | |||||||||
| Hazard Codes | Xi | |||||||||
| Risk Statements | 36/37/38 | |||||||||
| Safety Statements | 26-36-37/39 | |||||||||
| RIDADR | UN 3335 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | AJ2430000 | |||||||||
| F | 13 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 29163400 | |||||||||
| Toxicity | LD50 orally in Rabbit: > 5000 mg/kg LD50 dermal Rabbit > 5000 mg/kg | |||||||||
| NFPA 704 |
|
Phenylacetic acid Chemical Properties,Uses,Production
Chemical Properties
Phenylacetic Acid occurs in Japanese peppermint
oil, in neroli oil, and in traces in rose oils. It is a volatile aroma constituent
of many foods (e.g., honey). It forms colorless crystals (mp 78°C) that have a
honey odor.
The common route to phenylacetic acid is conversion of benzyl chloride into
benzyl cyanide by reaction with sodium cyanide, followed by hydrolysis. Because of its intense odor, phenylacetic acid is added to perfumes in small quantities
for rounding off blossom odors. Addition to fruit aromas imparts a sweet
honey note.
Occurrence
Reported found among the constituents of a few essential oils: tobacco, Rosa centifolia, Bulgarian rose, orange flowers absolute, neroli and Mentha arvensis of Japanese origin; also reported present among the volatile constituents of cocoa. Also reported found in guava, papaya, raspberry, strawberry, cooked potato, tomato, peppermint oil, pepper, rye bread, cheddar cheese, Swiss cheese, Gruyere cheese, boiled mutton, beer, cognac, cider, sherry, grape wines, white wine, sake, cocoa, tea, honey soy protein, passion fruit, starfruit, mango, mushroom, malt, wort, roasted chicory root, naranjilla fruit, choke berry, sea buckthorn and Chinese quince.
Uses
Phenylacetic Acid is used in the synthesis of Diclofenac (D436450) and its metabolite 4'-Hydroxydiclofenac (H825225), which is the principal human metabolite of Diclofenac.
Preparation
By the treatment of benzyl cyanide with dilute sulfuric acid and other processes.
Definition
ChEBI: A monocarboxylic acid that is toluene in which one of the hydrogens of the methyl group has been replaced by a carboxy group.
Safety Profile
Moderately toxic by ingestion, subcutaneous, and intraperitoneal routes. An experimental teratogen. Combustible liquid. Used in production of drugs of abuse. When heated to decomposition it emits acrid smoke and irritating fumes
Metabolism
Phenylacetic acid is conjugated in man and the chimpanzee, but probably in no other species, with glutamine. In most other animals, except the hen, it behaves like benzoic acid, forming glycine and glucuronic acid conjugates. In the hen, it conjugates with ornithine, forming phenacetornithuric acid. Phenacetylglutamine and its addition compound with urea were isolated from human urine alter the administration of phenylacetic acid (Williams, 1959).
Purification Methods
Crystallise the acid from pet ether (b 40-60o), isopropyl alcohol, 50% aqueous EtOH or hot water (m 77.8-78.2o). Dry it in vacuo. It can be distilled under a vacuum. [Beilstein 9 II 294, 9 III 2169.]
Phenylacetic acid Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
1of7
chevron_right




